To help develop antiviral drugs, Graduate Student Lucy Fallon in Carlos Simmerling’s lab is studying how the SARS-CoV-2 Spike protein binds with the host cell’s receptor, and models how different molecules might interfere with this interaction. She is also a gifted molecular-graphics artist. Here are some of her images that illustrate her project, and some other proteins as well.
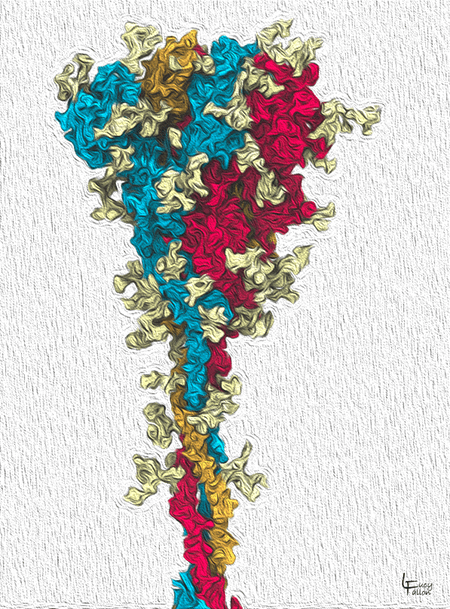
The SARS-CoV-2 Spike glycoprotein
CoV-2 Spike proteins reach out from the virus membrane and attach to receptors on host cells to invade them. The Spike is made from 3 identical protein chains (red, orange, blue). The light yellow protrusions on its surface are glycans, sugars that shield the protein from the host’s immune system. Notice that all 3 chains are packed together at the top. One chain will open up to bind to the receptor, as shown in figures below.
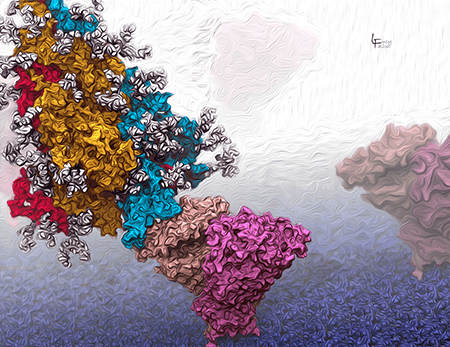
The SARS-CoV-2 Spike glycoprotein binding to ACE2
The Spike protein (red, orange, blue, with white glycans) binds to the host ACE2 (pink) receptor, which is embedded in the host cell membrane (violet blue, at the bottom). Here, the Spike is in the ‘open’, or active form in which the blue chain extends to bind ACE2.
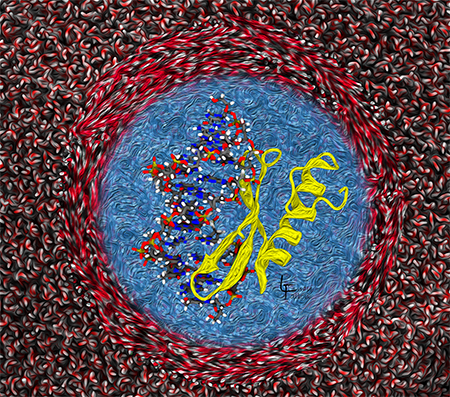
Implicit solvation: simplifying the representation of water
The center shows a protein-DNA complex immersed in a blue ‘field’ of water. This blue field represents implicit solvation, where the water is calculated as a dielectric field, making it possible to simulate the complex behavior in a reasonable amount of time. The outer region represents an alternative way an alternative method that takes a lot of time to calculate the positions and velocities of each water molecule.
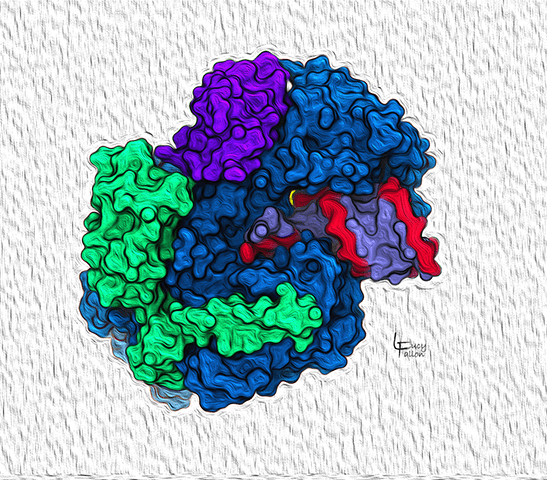
SARS-CoV-2 RNA-dependent RNA polymerase (RDRP) complex
A functional SARS-CoV-2 RDRP complex is composed of proteins that exist individually without defined structures, but associate to form the complexes shown here. The blue protein binds one complete RNA chain and links RNA bases to form a second matching RNA chain (red and blue-violet), which carry the virus’s genetic code into the next generation of virions. The green and deep purple proteins help stabilize the working mechanism.
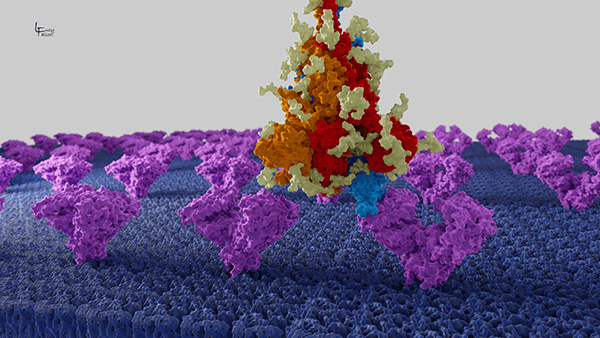
The SARS-CoV-2 Spike glycoprotein binding to ACE2
Here we look from a distance at the CoV-2 Spike protein (red, orange, blue with light yellow glycans) as it binds to the ACE2 receptor (purple) embedded in the host cell membrane (blue).
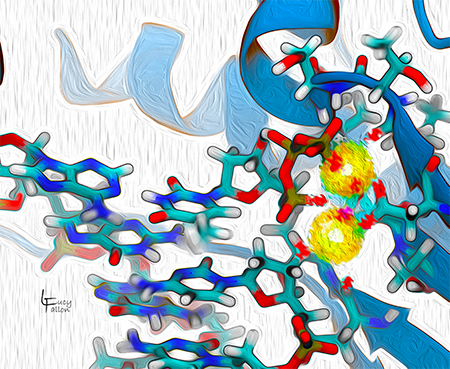
DNA Polymerase active site in the process of replicating DNA
DNA polymerase is responsible for faithfully replicating DNA chains in most organisms and some viruses. Using one DNA strand as a template, it links DNA bases to form a matching DNA chain. The active site is where the chemistry for lengthening the chain takes place. It uses two magnesium ions (yellow) to catalyze the reaction.
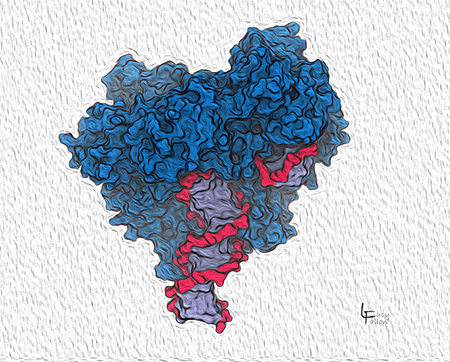
Surface view of DNA Polymerase
Here we see how DNA Polymerase (blue) grips DNA to position it on the active site. The DNA is shown with a red backbone and blue-violet bases in the center. This DNA is a double helix of the product daughter and the template strands. The active site is buried in this view.
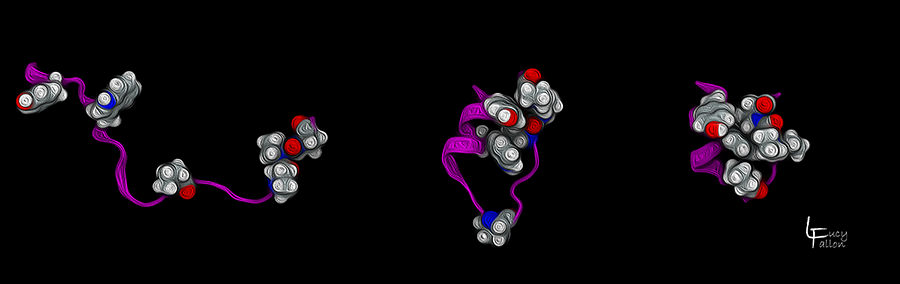
TrpCage Protein Folding
Here is one of the Simmerling Lab’s favorite model proteins: TrpCage (pronounced “trip cage”), a small protein that folds into a distinct, well-defined shape. It begins in its extended, unfolded form (left), then collapses its core residues (shown as red and white atoms) on top of each other (middle). Finally, it folds into its native functional state (right), where the core residues stack on top of each other to form a ‘cage’.